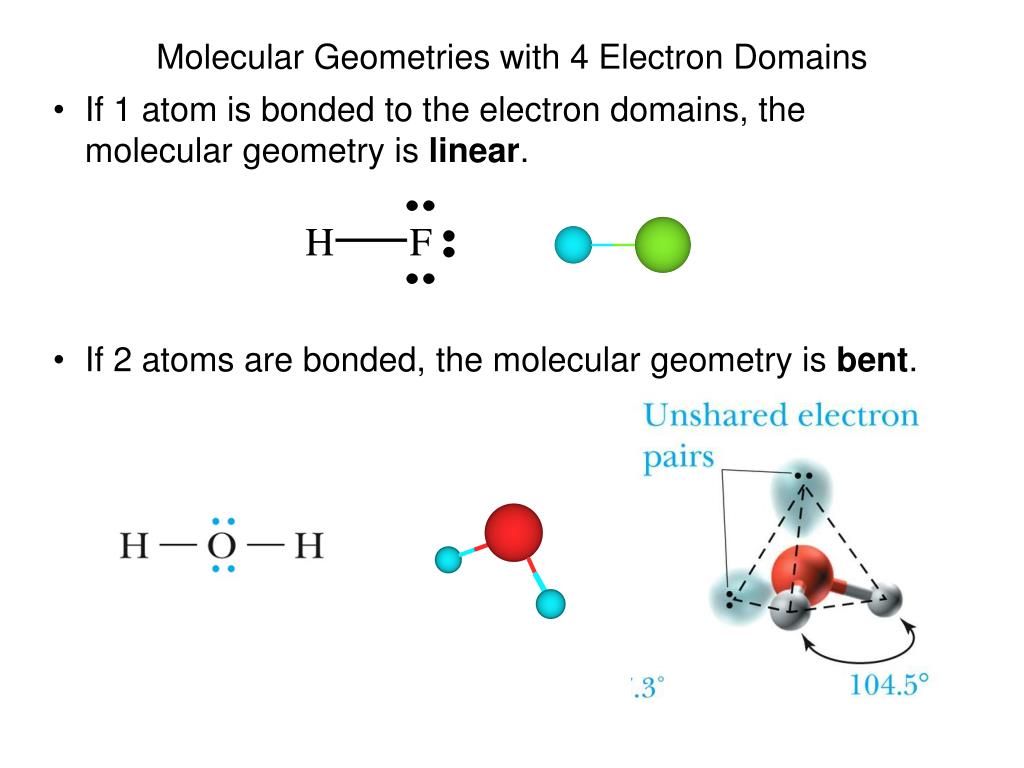
* Lone electron pairs are represented by a line without an atom attached. The table below shows the electron pair geometries for the structures we've been looking at: The regions of electron density will arrange themselves around the central atom so that they are as far apart from each other as possible. Use this number to determine the electron pair geometry.Įach bond (whether it be a single, double or triple bond) and each lone electron pair is a region of electron density around the central atom. Step 3: Add these two numbers together to get the regions of electron density around the central atom. Make sure you understand why they are correct. The correct answers have been entered for you. How many lone electron pairs are on the central atom in each of the following Lewis structures?  
Step 1: Count the number of lone electron pairs on the central atom. Molecular geometry can be predicted using VSEPR by following a series of steps: In other words, the electrons will try to be as far apart as possible while still bonded to the central atom. The theory says that repulsion among the pairs of electrons on a central atom (whether bonding or non-bonding electron pairs) will control the geometry of the molecule. The valence shell electron-pair repulsion theory (abbreviated VSEPR) is commonly used to predict molecular geometry. Lewis structures are very useful in predicting the geometry of a molecule or ion. Moore, John W., Stanitski, Conrad L., Wood, James L., Kotz, John C., The Chemical World, Concepts and Applications, Second Edition, Volume 1, Harcourt Brace & Co., 1998, pages 397-447.Many of the physical and chemical properties of a molecule or ion are determined by its three-dimensional shape (or molecular geometry). Ebbing, Darrell D., Wrighton, Mark S., General Chemistry, Second Edition, Houghton Mifflin Co., 1987 pages 286-321.Ģ. To predict the molecular geometry select from the table below the 3D arrangement that has the same number of bond domainsand lone pairs of electrons.ġ. Determine the number of bond domains and the number of lone pairs of electrons.ģ. Halogens and noble gases can expand their octet.Ģ. Note: To chose the central atom as the one with the smallest number of valence electrons or if they all have the same number of valence electrons then choose the one in the least amount. Draw Lewis structures for the molecular formula given. How to use the table to predict molecular geometry.ġ. For example, the molecule ethylene, H 2C=CH 2, has the carbon-carbon atoms sharing four electrons but one bond domain between the two carbon atoms. In any case, if two atoms share two electrons or more they will have one bond domain. For ease of understanding we will call a bond domain the electrons that hold together two atoms. The following table will help you understand how molecular geometry can be predicted using the VSPER model. On the other hand it takes into account the very important Pauli exclusion principle where each electron pair must occupy a different spatial region about an atom. On the first hand it minimizes repulsion between electrons due to electrostatic interactions. The VSPER model is based on two important principles. This model proposes that electrons are arranged around atoms in pairs such that they are kept as far away as possible. The valence shell electron pair repulsion (VSPER pronounced "vesper") model provides some useful tools for predicting molecular geometries. Atoms have a definite three-dimensional spacearrangement relative to each other in a molecule. can also be observed in the microscopic world. The geometrical arrangements seen in nature, i.e., flowers,stones, trees, etc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
